What is the oxygen dissociation curve?
The oxygen dissociation curve is a graph that plots the proportion of haemoglobin in its oxygen-laden saturated form on the vertical axis against the partial pressure of oxygen on the horizontal axis. The curve is a valuable aid in understanding how the blood carries and releases oxygen and it is a common theme that is tested on in many medical examinations.
At high partial pressures of oxygen, haemoglobin binds to oxygen to form oxyhaemoglobin. All of the red blood cells are in the form of oxyhaemoglobin when the blood is fully saturated with oxygen. Each gram of haemoglobin can combine with 1.34 mL of oxygen. At low partial pressures of oxygen (e.g. within tissues that are deprived of oxygen), oxyhaemoglobin releases the oxygen to form haemoglobin.
The oxygen dissociation curve has a sigmoid shape because of the co-operative binding of oxygen to the 4 polypeptide chains. Co-operative binding means that haemoglobin has a greater ability to bind oxygen after a subunit has already bound oxygen. Haemoglobin is, therefore, most attracted to oxygen when 3 of the 4 polypeptide chains are bound to oxygen.
There is often a P50 value expressed on the curve, which is the value that tells us the partial pressure of oxygen at which the red blood cells are 50% saturated with oxygen. At an oxygen saturation of 50%, the PaO2 is approximately 25 mmHg (3.5k Pa).
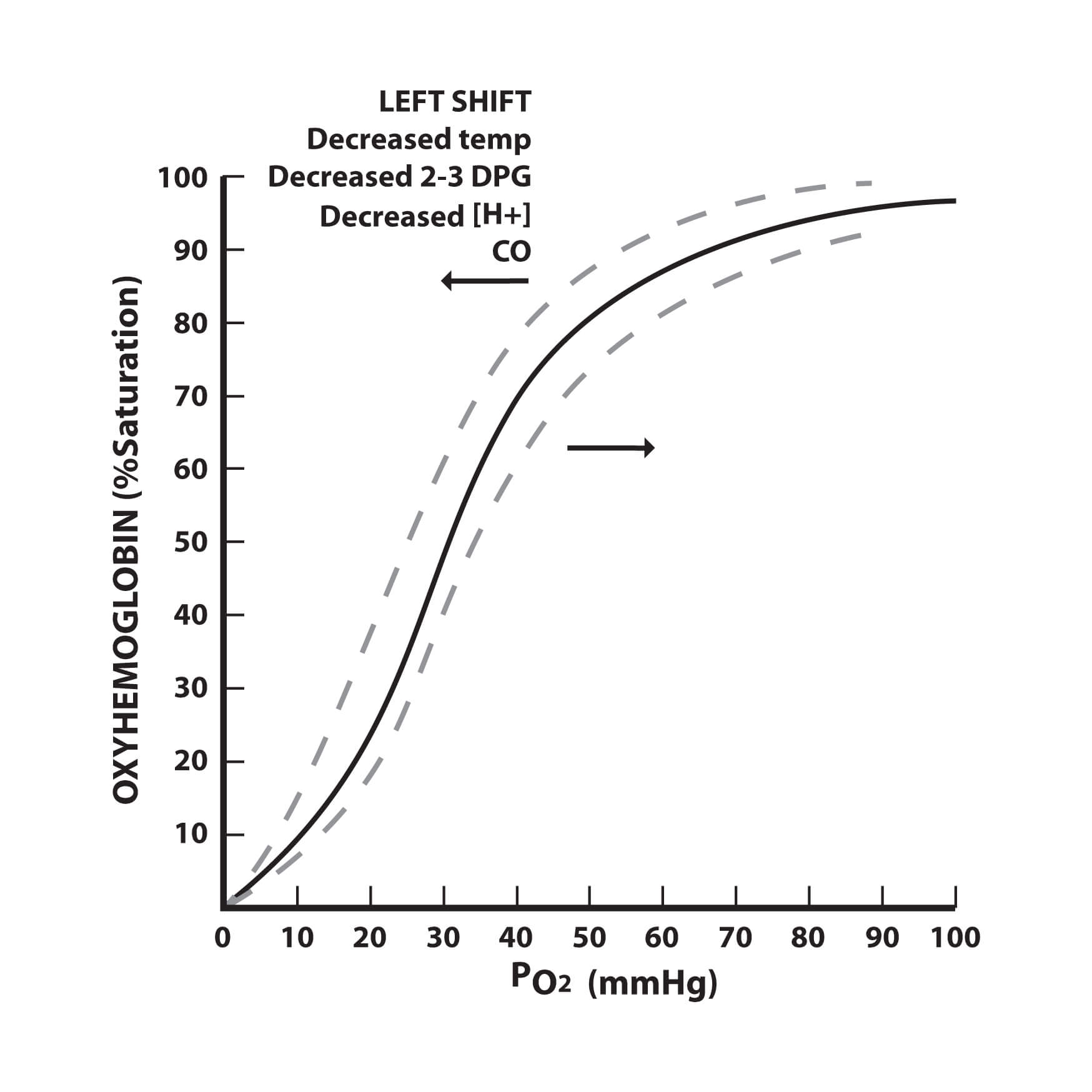
The oxygen dissociation curve and the factors affecting it.
Which factors affect the oxygen dissociation curve?
The oxygen dissociation curve can be shifted right or left by a variety of factors. A right shift indicates decreased oxygen affinity of haemoglobin allowing more oxygen to be available to the tissues. A left shift indicates increased oxygen affinity of haemoglobin allowing less oxygen to be available to the tissues.
pH:
A decrease in the pH shifts the curve to the right, while an increase in pH shifts the curve to the left. This occurs because a higher hydrogen ion concentration causes an alteration in amino acid residues that stabilises deoxyhaemoglobin in a state (the T state) that has a lower affinity for oxygen. This rightward shift is referred to as the Bohr effect.
Carbon dioxide (CO2):
A decrease in CO2 shifts the curve to the left, while an increase in CO2 shifts the curve to the right. CO2 affects the curve in two ways. Firstly, the accumulation of CO2 causes carbamino compounds to be generated, which bind to oxygen and form carbaminohaemoglobin. Carbaminohaemoglobin stabilizes deoxyhaemoglobin in the T state. Secondly, the accumulation of CO2 causes an increase in H+ ion concentrations and a decrease in the pH, which will shift the curve to the right as explained above.
Temperature:
An increase in temperature shifts the curve to the right, whilst a decrease in temperature shifts the curve to the left. Increasing the temperature denatures the bond between oxygen and haemoglobin, which increases the amount of oxygen and haemoglobin and decreases the concentration of oxyhaemoglobin. Temperature does not have a dramatic effect but the effects are noticeable in cases of hypothermia and hyperthermia.
Organic phosphates:
2,3-Diphosphoglycerate (2,3-DPG) is the main primary organic phosphate. An increase in 2,3-DPG shifts the curve to the right, whilst a decrease in 2,3-DPG shifts the curve to the left. 2,3-DPG binds to haemoglobin and rearranges it into the T state, which decreases its affinity for oxygen.
A table summarizing these effects is shown below:
| Factor | Decrease | Increase |
|---|---|---|
| pH | Right shift | Left shift |
| CO2 | Left shift | Right shift |
| Temperature | Left shift | Right shift |
| 2,3-DPG | Left shift | Right shift |
How does carbon monoxide affect the curve?
Carbon monoxide (CO) interferes with the oxygen transport function of the blood by combining with haemoglobin to form carboxyhaemoglobin (COHb). CO has approximately 240 times the affinity for haemoglobin than oxygen does and for that reason, even small amounts of CO can tie up a large proportion of the haemoglobin in the blood making it unavailable for oxygen carriage. If this happens the PO2 of the blood and haemoglobin concentration will be normal but the oxygen concentration will be grossly reduced. The presence of COHb also causes the oxygen dissociation curve to be shifted to the left, interfering with the unloading of oxygen.
How does methaemoglobin affect the curve?
Methaemoglobin is an abnormal form of haemoglobin in which the normal ferrous form is converted to the ferric state. Methaemoglobinaemia causes a left shift in the curve as methaemoglobin does not unload oxygen from haemoglobin.
The other oxygen transport molecules
There are two other oxygen transport molecules that are required knowledge and commonly asked about in medical exams, fetal haemoglobin and myoglobin:
Fetal haemoglobin
Fetal haemoglobin (HbF) is the main oxygen transport protein in the human fetus during the last 7 months of development. It persists in the newborn until roughly 6 months of age. HbF has different globin chains to adult haemoglobin (Hb). Whereas adult haemoglobin is composed of two alpha and two beta subunits, fetal haemoglobin is composed of two alpha and two gamma subunits. This change in the globin chain results in a greater affinity for oxygen and allows the fetus to extract oxygen from the maternal circulation. This increased affinity for oxygen means that the oxygen dissociation curve for fetal haemoglobin is shifted to the left of that of adult haemoglobin.
Myoglobin:
The curve for myoglobin lies even further to the left than that of fetal haemoglobin and has a hyperbolic, not sigmoidal, shape. Myoglobin has a very high affinity for oxygen and acts as an oxygen storage molecule. It only releases oxygen when the partial pressure of oxygen has fallen considerably. The function of myoglobin is to provide additional oxygen to muscles during periods of anaerobic respiration.
Header image used on licence from Shutterstock
Thank you to the joint editorial team of www.mrcemexamprep.net for this article.






very informative and useful review.
Helpful stuff
Amazing information
Great stuff- thank you!
SO GOOD
Concept clearly understood.
Simple and infomative
This is very informative and straight foward, yet exclusive. Cant thank you enough!
Simple, concise, precise and informative.
very good
Great information!
I never understood till now
Nice information
Concept is clearly explained and easy to understand.
easy to understand
Meaningful content
Thanks you sooooooooo much
Comment. great info
Very informative and precise. Very helpful. Thank you
This information was broken down in the most simplest way possible to make it so easy to understand and teach to others. My tendency is to focus on and overemphasize certain areas that may be easy for me to understand, but overlook other factors that are as equally important to notice and cover as well.
I owe you one for this. Great write up.
Great write..super informative..keep it up
How I wish this was how it was taught in Med school…but then again, I am older now, and can understand much better with a more mature level of understanding. Nevertheless, well explained! Thank you.
great information here..
Cheers mate, some very helpful information
God bless you
quite informative in a simpler manner
Well explained. I like this
Awesome read
Well explained
Easy to grasp
Better explanation, thx
Very well explained. Thank you
Well, well, well what a clear, simple and understandable info.
WELL RESEARCH DATA, VERY USEFUL INFORMATION
Fantastic work… Simple and very well explained… Thank you
Very helpful thank you🙏
Well simplified work, thank you and God bless
Helpful. Thank You
For the first time i didnt get any difficulty in grasping science.
thanks, produce more
So lovely and enjoyable thanks
EASY TO UNDERSTAND
very enjoyable